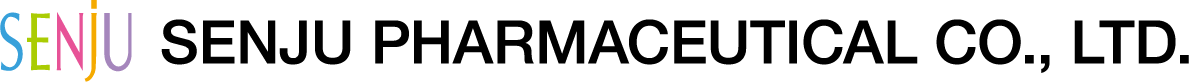This medicine is recommended for people who have continuing allergy symptoms with inflammation.

Category 2 OTC medicines
Mytear® ALPITATTO NEXα
Works on unbearable eye itching due to pollens and other reasons
CHARACTERISTICS
Allergy symptoms to pollen, etc. can cause inflammation of the eyes.
Then, inflammation not only causes the symptom of itchy eyes, but also worsens symptoms such as foreign body sensation and hyperemia (makes the symptoms feel worse).
- This product exerts its effect on inflammatory allergic symptoms (itching and foreign-body sensation [gritty and tingling sensations]) and reddening due to pollens and other reasons with its 3 active ingredients※1.
- It also contains a cornea-protecting ingredient※2.
- Mild but not cool sensation eye drop
※2 Sodium chondroitin sulfate
PRECAUTIONS FOR USE
What you should not do
(If you do not follow the precautions listed below, your current symptoms may worsen and you may be at an increased risk of side effects and accidents.)
1. The following persons should not use this medicine:
- Children under 7 years
- Women who are or may be pregnant
- Women who are breastfeeding
2. Do not operate vehicles or machines if you use this medicine with nasal drops. (Sleepiness may appear.)
Who should seek consultation
1. The following persons should consult a physician, pharmacist, or registered salesperson* before using this medicine:
- Persons under treatment by a physician
- Persons under treatment of allergy such as hyposensitization therapy
- Persons who have had allergies to medicines or something
- Persons who have suffered from the following symptom: Severe eye pain
- Persons diagnosed with the following condition: Glaucoma
- Persons who do not know whether their symptoms are caused by allergy or by other causes. Especially, persons in the following cases should consult a physician before using this medicine, because you cannot judge if the symptoms are due to allergy:
・If you have symptoms in only one eye.
・If you have symptoms in only the eyes and not in the nose.
・If your eyesight is affected.
2. Stop using this medicine immediately and consult a physician, pharmacist, or registered salesperson if the following symptoms appear after using this medicine because they might be side effects of the medicine. Take this leaflet with you.
| Affected area | Symptoms |
|---|---|
| Skin | Rashes, redness, itching |
| Eye | Hyperemia, itching, swelling (including around the eyes), irritation, pain, foreign body sensation, watery eyes, eye mucus |
| Others | Breathing difficulties |
In rare cases, serious symptoms might occur. In such a case, immediately consult a physician.
| Name of the symptom | Symptoms |
|---|---|
| Anaphylaxis | Symptoms such as breathing difficulties, edema (for example, swelling of the throat, eye lids, nasal membrane, and lips), and urticaria appear soon after using this medicine. |
3. Stop using this medicine immediately and consult a physician, pharmacist, or registered salesperson in the following cases. Take this leaflet with you.
- If your symptoms have worsen.
- If there is no improvement in your blurred vision. (You may have other disease such as glaucoma.)
- If there is no improvement in your symptoms after using this medicine for 2 days.
- If you use this medicine for more than 2 weeks even after there is an improvement.
INDICATIONS
Relief of the following allergy symptoms in the eyes caused by pollen, house dust, etc.:
Conjunctival hyperemia, itchy eyes, blurred vision (eye mucus), teary eye, foreign body sensation (gritty sensation)
DOSAGE AND DIRECTIONS
Apply 1 to 2 drops at a time, 4 times a day.
< Precautions regarding dosage and directions >
-
- Children are permitted to use this medicine only under the direction and supervision of a parent or other responsible adult.
- Do not touch the tip of the bottle to your eye, eyelid, or eye lashes. (If eye mucus or other foreign matter, etc. is mixed in the bottle, the medical solution can be contaminated or clouded.) In addition, do not use a clouded solution.
- Do not use this medicine while wearing contact lenses. (Take off your contact lenses once and apply.)
- For ophthalmic use only.
- Please follow the recommended dosage and directions.
-
INGREDIENTS
| INGREDIENTS | CONTENTS(in 1mL) |
|---|---|
| Sodium cromoglicate | 10mg |
| Chlorpheniramine maleate | 0.3mg |
| Pranoprofen | 0.5mg |
| Sodium chondroitin sulfate | 5mg |
Inactive ingredients
Boric acid, disodium edetate hydrate, dibutylhydroxytoluene, benzalkonium chloride, polysorbate 80, sodium borate, pH control chemicals
PRECAUTIONS FOR STORAGE AND HANDLING
-
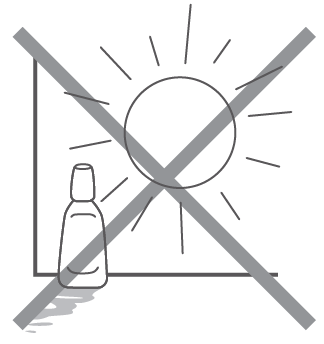
- Keep the bottle tightly closed and store it in a cool place away from direct sunlight. Do not leave the medicine where the temperature might become high (especially in cars or near heating appliances).
- In order to prevent changes in quality of the medicine by light, keep away from light before use, for example keep the bottle in this box.
- Keep out of the reach of children.
- To prevent improper use or loss of quality, do not transfer the medicine to another container.
- Do not transfer other things to the bottle.
- Do not share the medicine with others.
- Do not use this medicine after the expiration date. Even before the expiration date, use this medicine as soon as possible once the inner pouch is opened.
- Depending on the conditions of storage, ingredients of the medical solution can be crystalized and attached around the tip of the bottle or inside the cap. In such a case, wipe lightly with clean gauze before use.
-
| PACKAGE | 15mL |
|---|